Solve
Guides
0
Question
Which of the following is not linear?
- CO2
- H2O
- HCN
- C2H2
Open in App
Solution
Verified by Toppr
CO2, HCN, C2H2 are linear in shape.O=C=O,H−C≡N,HC≡CHH2O is bent or 'V' in shape.
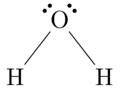
So, the answer is option D.
H2O is bent or 'V' in shape.
So, the answer is option D.
Was this answer helpful?
0