We all know very well what is heat and what it is capable of doing. But, specific heat is a concept that very few people know about. Also, we use specific heat to raise the temperature of an object by degree. In addition, we will discuss specific heat, specific heat formula, its derivation and solved examples in this topic. Furthermore, the specific heat of different elements differs from each other.
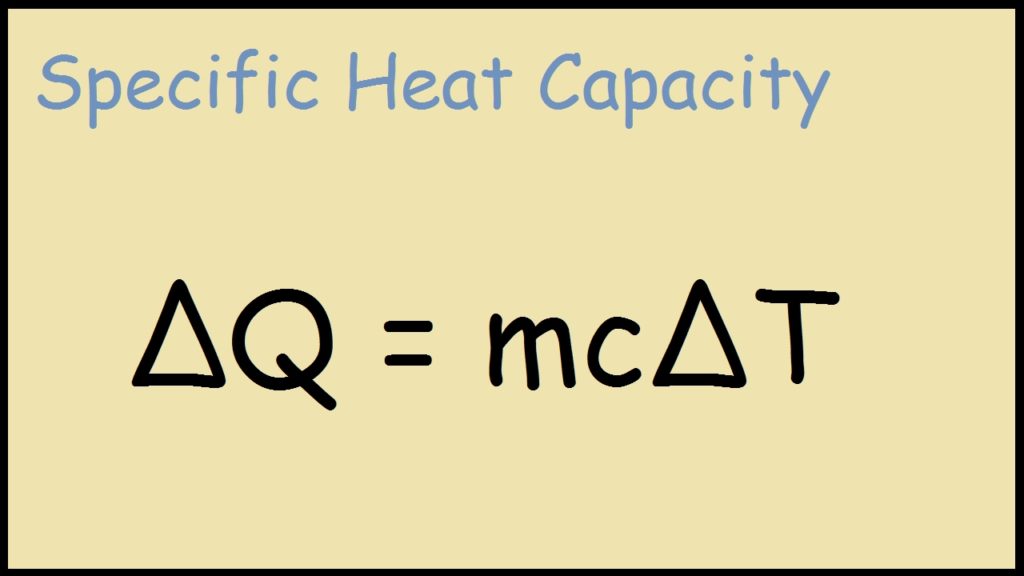
Source: YouTube
Specific Heat
Specific heat refers to the ratio of the quantity of heat that we require to raise the temperature of a body by one degree that we need to increase the temperature of an equivalent mass of liquid (water) by one degree. Also, we use the term in a narrower sense to mean the amount of heat, in calories that we require to raise the temperature of one gram of a substance by one Celsius degree.
In simple words, it is the ratio of the amount of heat required to increase the temperature of an object by one degree to the amount of heat required to increase the temperature of the same amount of water by one degree.
Get the huge list of Physics Formulas here
Specific Heat Formula
As we discussed above the specific heat is the relation of temperature change of an object with water.
Also, the formula is like this:
Heat energy = (mass of the object or substance) × (specific heat) × (Change in temperature)
Q = m × c × \(\Delta T\)
Or
Q = \(mc\Delta T\)
Derivation of Specific Heat Formula
Q = refers to the heat energy in Joules (J)
m = refers to the mass of the substance in kilogram (kg)
c = refers to the specific heat in joules per kilogram (\(J/kg\cdot k\))
\(\Delta\) = refers to the symbol of change
\(\Delta t\) = refers to the change in temperature in kelvins (K)
Solved Examples on Specific Heat Formula
Example 1
If the specific heat of gold is 129 \(J/kg\cdot k\). Then what quantity of heat energy is required to raise the temperature of 100 g of gold by 50.0 K?
Solution:
First of all, write down the things given in the question
Mass of the gold = 100 g converting it into kg, we get 0.100 kg.
Specific heat = 129 \(J/kg\cdot k\).
Temperature = 50.0 K
Calculation:
Q = \(mc\Delta T\)
Q = (0.100 kg) (129 \(J/kg\cdot k\)) (50.0 K)
So, the energy required to raise the temperature of 100 g gold is 645 J.
Example 2
Suppose a pot is heated by transferring 1676 KJ of heat energy to the water. Moreover, if there is 5.000 kg of water in the pot and the temperature is raised by 80.0 K then find the specific heat of water?
Solution:
Let’s write down the things given in the question
Mass (m) = 5.00 kg
Temperature (T) = 80.0 K
Specific heat (c) = 1676 KJ
Now we have to convert the specific heat into Joules because it is in Kilojoules.
So, the conversion is like this
1 KJ = 1,000 J
So, 1676 KJ = 1,000 × 1676 = 16,76,000 J
Now put all the values in the formula. But, before that, we have to reorganize the formula to find specific heat.
Q = \(mc\Delta T\) \(\rightarrow\) c = \(\frac{Q}{m\Delta T}\)
Now putting values in the rearranged formula
C = \(\frac{1676000} {(5.00 kg)(80.0)}\)
C = 4190 \(J/kg\cdot k\)
Hence, the specific heat of water is 4190 \(J/kg\cdot k\).






Typo Error>
Speed of Light, C = 299,792,458 m/s in vacuum
So U s/b C = 3 x 10^8 m/s
Not that C = 3 x 108 m/s
to imply C = 324 m/s
A bullet is faster than 324m/s
I have realy intrested to to this topic
m=f/a correct this
Interesting studies
It is already correct f= ma by second newton formula…